The violations of regulatory compliance may lead to the issue of a Warning Letter FDA WARNING LETTER AND FDA FORM 483. Over the recent years, most of these warning letters are due to non-compliance or negligence in the audit trail documentation practices.
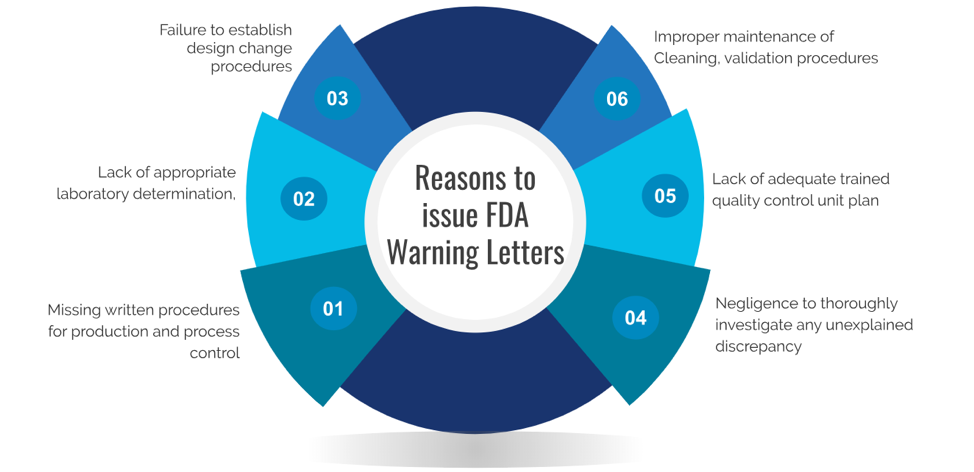
Most frequent reasons to Issue FDA warning letters
Document control best practices to help companies avoid FDA warning letters
- Establish Document Control Procedures: The FDA commonly issues warning letters over the lack of document control procedures:
- The means no backdating records, which the agency has noted as a problem in the past.
- The old copies of SOPs taped up next to a workstation.
- Permissions need to be set up that prevent unauthorized individuals from changing key documents such as SOPs, specifications and testing methods.
- Integrate Document Control with Quality Processes: The written procedures should be effectively integrated document Control systems and procedures into their larger quality process.
- Protect Data Integrity: The FDA expects data to be attributable, legible, complete, an original or true copy and accurate. Essential document control practices for protecting data integrity include:
- Retaining original records or true copies.
- Documenting all data from every test.
- Real-time data recording.
An automated QMS can help protect data integrity by pulling the research results data directly from the LIMS.
- Control Paper Records: Best practices for controlling paper records include:
- Treating forms, worksheets, and notebooks as controlled documents.
- Making sure all blank forms issued are numbered and reconciled.
- Retaining incomplete forms and recording why they were replaced.
- Identifying unofficial notebooks, stamping notebooks for official use and inspecting pages for gaps.
Cloudbyz CTMS Solutions integrated with Documents control best practices to avoid FDA warning letters
- Be Inspection Ready: Cloudbyz CTMS meets operational needs and compliance requirements, the audits trial features are enabled that helps to conduct internal and external audits on a regular basis. Cloudbyz CTMS enabled to inform the employees about audits, resources needed and staff recruitment to accompany auditors through email communication.
- eTMF for enhanced documentation system: The eTMF feature in Cloudbyz CTMS enables to store the essential documents with a click of a button and through drag & drop facility. The folders can be structured according to the DIA or customized, with audit tracking and automated document approval process hassle-free.
- Access controlled documentation Feature: Cloudbyz CTMS ensures an automated document control system including SOPs with a proper version that eliminates the possibility of human errors. Cloudbyz CTMS also establishes proper permissions to prevent unauthorized access and changes the documents.
- Develop an audit trial compliance culture: Cloudbyz CTMS ensures simplified audit trail management in the initial meeting of an audit, during the audit process, and all subsequent communications with the FDA.
- A Hyper-Adaptive Quality Management Software: Cloudbyz CTMS ensures a modern and hyper-adaptive quality automation solution that will ensure compliance today and into the future that addresses new business and regulatory changes.
Abbreviations:
-
- FDA: Food and Drug Administration
- CTMS: Clinical Trial Management System
- SOP: Standard Operating Procedure
- QMS: Quality Management System
- LIMS: Laboratory Information Management System
- eTMF: Electronic Trial Master File
- DIA: Drug Information Association
References
https://www.biospace.com/article/fda-warning-letters-an-overview-of-2019-s-letters-so-far/
https://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/EnforcementActivitiesbyFDA/
https://blog.etq.com/avoid-fda-warning-letters-with-these-4-document-control-best-practices
https://www.intellect.com/blog/6-tips-to-avoid-483-letters
https://rescop.com/blog/2018/03/01/fda-warning-letters/
https://www.softwaresuggest.com/blog/document-management-best-practices/
https://www.fda.gov/ICECI/EnforcementActions/WarningLetters/ucm636172.htm
https://www.fda.gov/ICECI/EnforcementActions/WarningLetters/ucm635686.htm
https://www.fda.gov/ICECI/EnforcementActions/WarningLetters/ucm635684.ht
https://www.pharmaceuticalonline.com/doc/an-analysis-of-fda-fy-drug-gmp-warning-letters-0003


